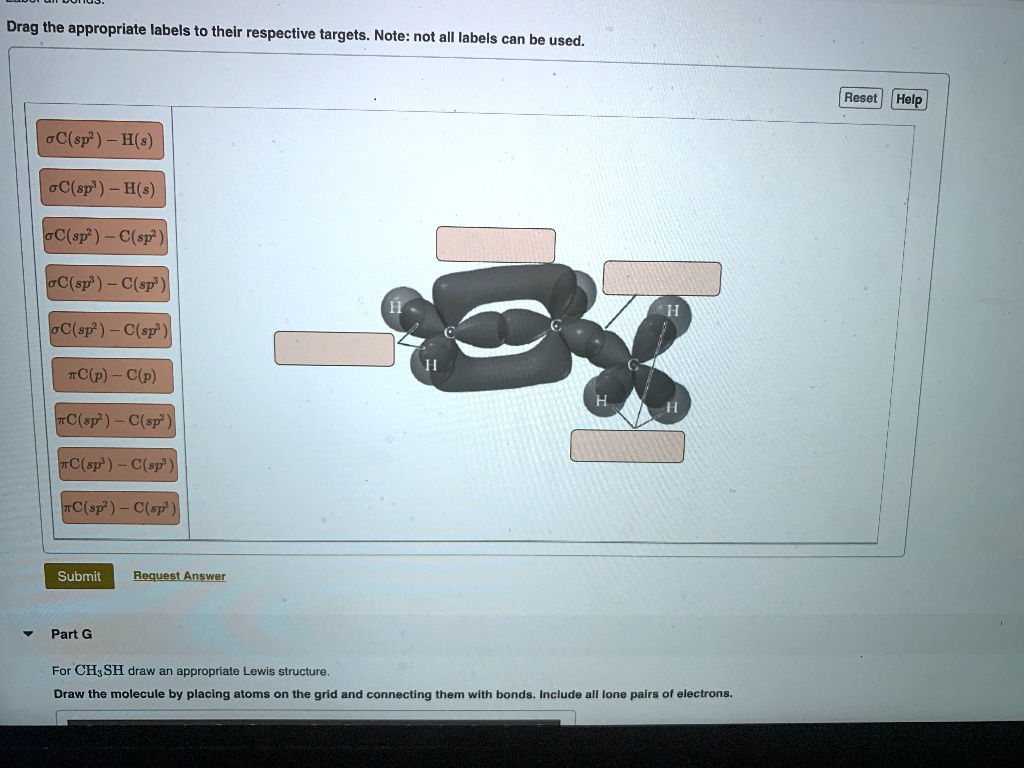
Drag the appropriate labels to their respective targets. Note: not all labels can be used. Reset Help oC(sp? ) H(s) oC(sp? H(s) loC(sp? ) C(sp? loC(sp" ) = C(sp'~ lC(sp? ) _ C(sps - #C(p) C(p) I~C(sp? ) C(sp? ~C(sp? ) C(sp' #C( sp? ) C(sp Submit Request Answer Part G For CHzSH draw an appropriate Lewis structure_ Draw the molecule by placing atoms on the grid and connecting them wlth bonds: Include all lone pairs of electrons: 3k
Expert's Answer
Here we've got the orbital model of a molecule that contains carbon bonded to 2, hydrogen double bonded to a carbon that is bonded to 1 hydrogen and another carbon which is bonded to 3. Hydrogen is, and it wants us to identify first what these bonds are well, each hydrogen has a 1 s bond that overlaps with a hybridized orbital 1 s; orbital, sorry that overlaps with a hybridized orbital carbon with 3 electron groups, that's s, p 2 hybridized, all single Bonds are sigma bonds, so this is a sigma carbon s, p, 2, to a hydrogen s, that's how we would describe these 2 bonds. The next 1 is the second bond, the pi bond associated with carbon carbon. So it's going to be a pie. Carbon p to a carbon p, which is an option and then the next 1 is this bond right here, it's a single bond! So it's a sigmal bond. We'Ve got a carbon to a carbon. This carbon is sp 2 hybridized with 3 electron groups, and this carbon is sp 3 hybridized with 4 electron groups and then last of all, it wants all of these they're single bonds. So they'll also be sigma. There'S a carbon with 4 electron groups. So it's sp. 3 hybridized bonding to a hydrogen that just has an s unhybridized atomic orbital. I hope that.
Chat with our Experts
Want to contact us directly? No Problem. We are always here for you

Your future, our responsibilty submit your task on time.
Order NowGet Online
Assignment Help Services





